藥品名稱
drug name | Influenza Vaccine (Vaxigrip) 0.25ml/syringe (巴斯德流感疫苗) 107年度公費流感疫苗 6個月以上,3歲以下 |
| 藥檔狀態 | |
成 份
Ingredient | Influenza virus vaccine |
| 單位含量 | 0.25mL/syringe |
| Dosage Forms | Suspension Prefilled Syringe, Intramuscular: 0.25mL |
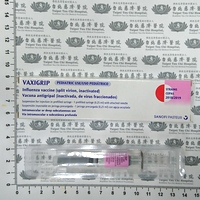
| 外觀描述 | 針劑/針筒 |
| Appearance | injection/syringe |
廠商名稱
Manufacturer | 賽諾菲股份有限公司 |
製 造 商
Manufacturer | SANOFI PASTEUR |
字 號
Product ID | 衛署菌疫輸字第000453號 |
藥理分類
Pharmacologic Category | Vaccine; Vaccine, Inactivated (Viral) |
作用機轉
Mechanism of action | Promotes immunity to seasonal influenza virus by inducing specific antibody production. Preparations from previous seasons must not be used. |
| 用途/適應症 | 流行性感冒疫苗 |
| Use |
Influenza disease prevention: Active immunization against influenza disease caused by influenza virus subtypes A and type B contained in the vaccine in the following persons:
US labeling:
‧ 6 months and older (Fluarix Quadrivalent, FluLaval Quadrivalent, Fluzone Quadrivalent)
‧ 4 years and older (Flucelvax Quadrivalent)
‧ 5 years and older (Afluria and Afluria Quadrivalent)
‧ 65 years and older (Fluad, Fluzone High-Dose)
Canadian labeling:
‧ 6 months to <2 years (Fluad Pediatric)
‧ 6 months and older (Agriflu, FluLaval Tetra, Fluviral, Fluzone Quadrivalent)
‧ 3 years and older (Influvac)
‧ 65 years and older (Fluad, Fluzone High-Dose)
The Advisory Committee on Immunization Practices (ACIP) recommends routine annual vaccination with the seasonal influenza vaccine for all persons≧6 months of age who do not otherwise have contraindications to the vaccine (ACIP [Grohskopf 2018]).
The ACIP recommends use of any age and risk factor appropriate product and does not have a preferential recommendation for use of the trivalent inactivated influenza vaccine (IIV3) or the quadrivalent inactivated influenza vaccine (IIV4). In addition to the IIV products, other alternative products are available for certain patient populations: Persons 18 years and older may receive vaccination with the recombinant influenza vaccine (RIV). Live attenuated influenza vaccine (LAIV4) is also an option for the 2018-2019 influenza season (CDC/ACIP [Grohskopf 2018]). Although ACIP does not express any preference for an influenza vaccine product, AAP recommends inactivated influenza vaccine as the preferred product for pediatric patients for the 2018-2019 season; live attenuated vaccine may be used if patient would not otherwise be vaccinated (eg, refusal of IIV3 or IIV4) and live vaccine is appropriate (AAP 2018; CDC/ACIP [Grohskopf 2018]).
The Canadian National Advisory Committee on Immunization (NACI) recommends annual vaccination with seasonal influenza vaccine for all persons≧6 months who do not otherwise have contraindications to the vaccine. Healthy, nonpregnant persons aged 2 to 59 years may receive vaccination with the seasonal live, attenuated influenza vaccine (LAIV) (nasal spray). For persons 6 months to 17 years of age, NACI recommends quadrivalent inactivated influenza vaccine (QIV) (trivalent inactivated influenza vaccine [TIV] if QIV is not available) (NACI 2018).
When vaccine supply is limited, target groups for vaccination (those at higher risk of complications from influenza infection and their close contacts) include the following (CDC/ACIP [Grohskopf 2018]):
‧ All infants and children 6 to 59 months of age
‧ Persons≧50 years of age
‧ Infants, children, and adolescents (6 months to 18 years of age) who are receiving long-term aspirin or salicylate therapy, and therefore, may be at risk for developing Reye syndrome after influenza
‧ Women who are or will be pregnant during the influenza season
‧ Patients with chronic pulmonary disorders (including asthma) or cardiovascular systems disorders (except isolated hypertension), renal, hepatic, neurologic, hematologic, or metabolic disorders (including diabetes mellitus)
‧ Persons who have immunosuppression due to any cause (including immunosuppression caused by medications or HIV)
‧ Residents of nursing homes and other long-term care facilities
‧ American Indians/Alaska Natives
‧ Morbidly obese (BMI≧40)
‧ Health care personnel including students in these professions who will have contact with patients
‧ Household contacts (including children) and caregivers of neonates, infants, and children <5 years (particularly neonates and infants <6 months) and adults≧50 years
‧ Household contacts (including children) and caregivers of persons with medical conditions which put them at higher risk of severe complications from influenza infection
In addition, the NACI also recommends vaccination of patients with neurologic or neurodevelopment conditions including neuromuscular/neurovascular/neurodegenerative conditions, seizure disorders (including febrile seizures in pediatric patients and isolated developmental delay) but excluding migraines and psychiatric conditions without neurological conditions (NACI 2018).
|
衛福部核准適用症狀
MOHW approved indications |
預防流感
|
| 常用劑量 | 立刻使用0.25mL
(藥品劑量會因人或病情增減,請依照醫師指示服用。) |
| Dose |
Pediatric:
It is important to note that influenza seasons vary in their timing and duration from year to year. In general, vaccination should begin soon after the vaccine becomes available such that the patient is fully immunized prior to onset of influenza activity in the community (eg, by end of October in US). However, vaccination should continue throughout the influenza season as long as vaccine is available. Unless noted, the ACIP and AAP do not have a preference for any given inactivated influenza vaccine (IIV) formulation when used within their specified age indications (AAP 2018; CDC/ACIP [Grohskopf 2018)]. International considerations: Products with similar names but containing different strains may be circulating globally due to differences in recommendations between northern and southern hemisphere countries. In addition, recommendations related to use of influenza vaccines and approved ages may vary per country.
仿單:
接種量:0.5歲以上未滿 3歲兒童:一劑0.25mL。
若先前未曾注射過流感疫苗者,需接種2次,且間隔至少4週。
Renal impairment:
There are no dosage adjustments provided in manufacturer`s labeling.
Hepatic impairment:
There are no dosage adjustments provided in manufacturer`s labeling.
|
懷孕分級
Pregnancy Risk Factor |
FDA:無。
兒童用,不適用懷孕分級
-----------------------------------------------------------
[FDA(美國食品及藥物管理局)懷孕分級說明:
A:對照試驗無法證實懷孕初期及後期使用會危害胎兒。
B:動物試驗無法證實對胎兒有害,但缺乏人類對照試驗;或動物試驗有副作用報告,但無法證實對懷孕初期及後期之胎兒有害。
C:動物實驗中對胎兒有害但缺乏孕婦對照實驗;或無動物及孕婦試驗。
D:證實對胎兒有害,但疾病對孕婦有生命威脅或較安全藥品無法使用或無效時可使用。
X:證實對胎兒有害,且使用後危害大於可能的益處。孕婦及可能懷孕婦女禁用。]
|
| 禁忌症 |
不適接種者:
先前接種本疫苗或對本疫苗任何成分曾發生嚴重過敏反應者。
注意事項:
(1) 發燒或正患有急性中重度疾病者,宜待病情穩定後再接種。
(2) 出生未滿6個月不適合接種。
(3) 先前接種本疫苗六週內曾發生格林-巴利症候群(Guillain-Barre syndrome, GBS)者。
(4) 已知對「蛋」之蛋白質有嚴重過敏者,可在門/住診由熟悉處理過敏症狀之醫事人員提供接種,並於接種後觀
察30分鐘,無不適症狀再離開。
(5) 其他經醫師評估不適合接種者。
|
| Contraindications |
Severe allergic reaction (eg, anaphylaxis) to a previous influenza vaccination or to any component of the formulation
Additional manufacturer contraindications for Afluria, Afluria Quadrivalent, Fluad, Fluarix Quadrivalent, FluLaval Quadrivalent, Fluzone High-Dose, Fluzone Quadrivalent: History of severe allergic reaction (eg, anaphylaxis) to egg protein
Additional manufacturer contraindications for Canadian products: Agriflu, Fluad, Fluad Pediatric, FluLaval Tetra, Fluviral, Fluzone High-Dose, Fluzone Quadrivalent, Influvac: Hypersensitivity to egg protein; hydrocortisone (Fluad and Fluad Pediatric only).
Note: Both ACIP and NACI do not consider egg allergy a contraindication to influenza vaccination (CDC/ACIP [Grohskopf 2018]; NACI 2018).
|
| 常見副作用 | 可能發生:咳嗽、嗜睡、腹瀉、食慾降低、發燒或注射部位疼痛。 |
| Common adverse drug reactions | Cough, sleepiness, diarrhea, decreased appetite, fever or injection site pain. |
| Adverse Reactions |
Frequency not defined. Adverse reactions in adults≧65 years of age may be greater using the high-dose vaccine, but are typically mild and transient.
Cardiovascular: Chest tightness, hypertension
Central nervous system: Chills, drowsiness, fatigue, headache, irritability, malaise, migraine, shivering
Dermatologic: Diaphoresis, ecchymoses
Gastrointestinal: Abdominal pain, anorexia, diarrhea, gastroenteritis, nausea, sore throat, vomiting
Infection: Infection, varicella
Local: Injection site reactions (including bruising, erythema, hematoma at injection site, induration, inflammation, itching at injection site, pain, rash, soreness, swelling at injection site, tenderness at injection site)
Neuromuscular & skeletal: Arthralgia, back pain, myalgia (may start within 6 to 12 hours and last 1 to 2 days; incidence generally equal to placebo in adults; occurs more frequently than placebo in children)
Respiratory: Bronchitis, cough, dyspnea, nasal congestion, nasopharyngitis, oropharyngeal pain, pharyngitis, pharyngolaryngeal pain, rhinitis, rhinorrhea, upper respiratory tract infection, wheezing
Miscellaneous: Fever
|
★高警訊藥品
監測建議 |
|
監測
Monitoring |
Monitor for anaphylaxis and syncope for 15 minutes following administration (ACIP [Kroger 2017]). If seizure-like activity associated with syncope occurs, maintain patient in supine or Trendelenburg position to reestablish adequate cerebral perfusion.
|
| 警語與注意事項 | 皮下或肌肉注射。可能會造成發燒或過敏反應。冷藏避光儲存。107.12.08公費流感疫苗為Vaxigrip。 |
| Warnings & precautions | For injection. Store in the refrigerator. Protect from light. |
| 針劑溶解條件 |
|
| 針劑稀釋條件 |
|
| 針劑不相容性 |
目前尚無相關臨床試驗數據或研究報告,因此不建議將本劑與其他廠牌季節性流感疫苗混用。(1071011仿單資料)
|
| 針劑施打條件 |
通常採皮下或肌肉注射。(1071011仿單資料)
|
| 針劑保存安定性 |
遮光儲存在 2~8℃,避免凍結。 (1071011仿單資料)
|
最近修改日期時間
Updated | 11/19/2019 4:58:47 PM |
|

現用藥品
Available
|
停用藥品
Old item
|

衛教單張
Drug info. handout
|

藥品仿單
DrugLabeling
|

二維條碼
QR code
|
|